 Since our inception, we have committed to innovation and continuously advanced our novel drug development. Through years of dedicated research in discovery, rapid clinical development and manufacturing improvement, we have built a mature portfolio of three commercialized innovative drugs, establishing a robust product portfolio and a continuous cash flow stream. This validated commercialization capability provides us with consistent momentum for business growth and lays a strong foundation for future market expansion and ongoing innovation.
Since our inception, we have committed to innovation and continuously advanced our novel drug development. Through years of dedicated research in discovery, rapid clinical development and manufacturing improvement, we have built a mature portfolio of three commercialized innovative drugs, establishing a robust product portfolio and a continuous cash flow stream. This validated commercialization capability provides us with consistent momentum for business growth and lays a strong foundation for future market expansion and ongoing innovation.
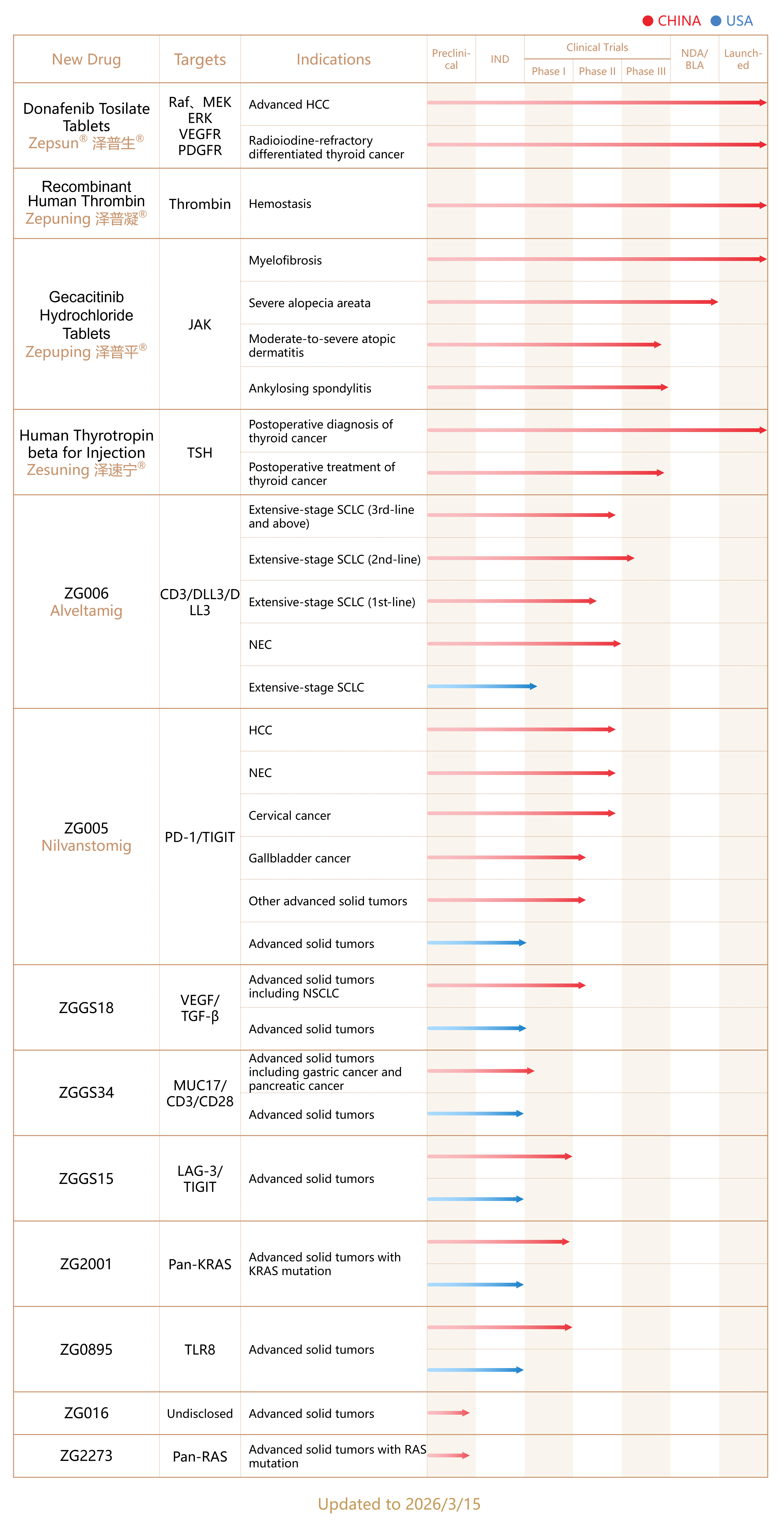
We have four marketed drugs: Zepsun® (Donafenib Tosilate Tablets), China's first domestically developed small molecule multi-target drug for the first-line treatment of advanced hepatocellular carcinoma; Zepuping® (Gecacitinib Hydrochloride Tablets), China's first domestically developed innovative JAK inhibitor for the treatment of myelofibrosis; Zepuning® (Recombinant Human Thrombin), the only recombinant human thrombin in China successfully commercialized using recombinant DNA technology; and Zesuning® (Human Thyrotropin Beta for Injection), China's first approved innovative product for postoperative precise assessment of differentiated thyroid cancer, used as a diagnostic drug for radioactive iodine whole-body scanning and serum thyroglobulin testing during postoperative follow-up for patients with differentiated thyroid cancer.
Zepsun® (Donafenib Tosilate Tablets). Zepsun® is the first domestically developed small-molecule multi-targeted drug approved in China in June 2021 for the first-line treatment of advanced HCC, based on the improved survival outcome in a Phase III clinical trial. Moreover, Zepsun® is the only monotherapy that has demonstrated superior survival outcome over sorafenib in a head-to-head clinical trial for advanced liver cancer. In August 2022, Zepsun® was subsequently approved for progressive, locally advanced or metastatic radioactive iodine-refractory differentiated thyroid cancer. Both indications have been included in the National Reimbursement Drug List (“NDRL”), which has supported the steady sales growth since launching. With its improved efficacy and overall favorable safety profile, it has been recommended as a first-line therapy in nearly 30 national treatment guidelines and consensus statements for liver and thyroid cancers. We have been continuously expanding the hospital and pharmacy coverage of Zepsun®, laying a strong foundation for sustained sales growth.
Zepuping ® (Gecacitinib Hydrochloride Tablets). Zepuping is the first domestically developed JAK inhibitor approved in China for the treatment of myelofibrosis. Approved for market launch in May 2025 for myelofibrosis, Zepuping is characterized by its distinct mechanism of action by targeting both JAK and Activin A Receptor Type 1 (“ACVR1”), thereby significantly improving its efficacy and safety. It has been recommended as a first-line treatment for myelofibrosis in the 2025 Chinese Society of Clinical Oncology (“CSCO”) Guidelines for Malignant Hematologic Diseases and is recommended as the preferred treatment for patients with concomitant anemia. In December 2025, Zepuping was approved for inclusion in the NRDL with effective date on January 1, 2026. Zepuping is also under active clinical development in multiple autoimmune indications with great market potential. Based on the positive Phase III results, its indication in severe alopecia is currently undergoing NDA review for commercialization approval, making it one of the most advanced domestic JAK inhibitors in this field. In addition, the Phase III clinical trial for ankylosing spondylitis has also reached its primary endpoint recently, while the Phase III trial for moderate to severe atopic dermatitis is ongoing, both representing potential significant market opportunities.
Zepuning (Recombinant Human Thrombin). Approved for market launch in January 2024 for commercialization, Zepuning is the only recombinant human thrombin developed with recombinant DNA technology and successfully commercialized in China. It has been recommended in the “Chinese Expert Consensus on Hemostasis in Hip and Knee Arthroplasty” by the Joint Surgery Group of the Orthopaedic Society of the Chinese Medical Association in 2025.
Zesuning® (Human Thyrotropin beta for Injection): Approved for market launch in January 2026, it is used as an diagnostic tool in the follow-up of patients with differentiated thyroid cancer after total or near-total thyroidectomy, for serum thyroglobulin (Tg) testing with or without radioactive iodine (¹³¹I) whole-body scan (WBS). This product fills the market gap in China for precise postoperative assessment of differentiated thyroid cancer.
As of December 2025, our strategically tiered pipeline of drug candidates comprises over 10 candidates covering nearly 30 major clinical projects (including Zepuping® and its related clinical programs in autoimmune diseases). Among these, seven indications across three candidates have progressed to the Biologics License Application (BLA)/New Drug Application (NDA) or pivotal/Phase III registration clinical trial stages. This includes the ongoing Phase III clinical trials for Zepuping® in severe alopecia areata, ankylosing spondylitis, and atopic dermatitis, which will expand its therapeutic scope into the field of autoimmune diseases.
We continue to invest in research and development targeting novel mechanisms and breakthrough technologies. Key projects include ZG006 (Alveltamig) (the world's first DLL3-targeting trispecific antibody with best-in-class potential) and ZG005 (Nilvanstomig) (a PD-1/TIGIT bispecific antibody, one of the most advanced programs globally, also possessing best-in-class and first-in-class potential). Particularly in oncology, we are developing innovative combination therapies to fully leverage the synergistic advantages of our product portfolio and R&D pipeline. We adopt a focused strategy to address the global unmet medical needs in refractory and recurrent cancers, including overcoming PD-1 resistance through combination regimens. Each of our core assets establishes a solid value foundation for global business expansion and partnership opportunities.
Furthermore, we are building a portfolio of cutting-edge early-stage projects, including ZGGS18, ZGGS34, ZGGS15, ZG2001, ZG0895, ZG016, and ZG2273. These cover T-cell engagers, bispecific and multispecific antibodies, as well as small molecule therapies targeting traditionally "undruggable" pathways. These projects exemplify our deep technical expertise and our ability to translate sustained scientific investment into breakthrough innovations.
With our continuously expanding pipeline and advancing late-stage programs, we are laying a solid foundation for future commercial growth. We remain committed to developing innovative drugs with global intellectual property rights that are safe, effective, and accessible, aiming to address significant unmet clinical needs both in China and worldwide.
The chart below summarizes the R&D progress of our marketed drugs and core drug candidates as of March 15th 2026: